IVF epigenetic damage gets worse across generations; The next Project Hail Mary; AI's "odorless" math proofs; Waymo at 100% human oversight? & more
Desiderata #39
The Desiderata series is a regular roundup of links and thoughts for paid subscribers, and an open thread for the community.
Contents
IVF epigenetic damage gets worse across generations
Waymo reveals their fleet may require 100% human oversight
The next Project Hail Mary
Nonfiction book sales drop twice as much as fiction
Art collective poisons AI training set
Lyme Disease vaccine announced (but is it better than prophylactic antibiotics?)
Terence Tao warns of “odorless” AI proofs that shed no insight
A terraformed mars might have Insects of Unusual Size
From the Archives: My worry of AI outputs surpassing human “outputs” comes true
Comment, share anything, ask anything
1. IVF epigenetic damage gets worse across generations
Last month there was an interesting paper in Nature Communications, “Limitations of serial cloning in mammals,” which showed that after 58 generations a cloned mice population had degraded genetically to the point where further cloning was impossible.
It’s a great argument for “Why sex?”
And it’s one very different from the “Red Queen” hypothesis I was taught in school (as basically gospel). The Red Queen hypothesis argues that sex is to shuffle around your genome to create diversity that evolution can capitalize on, especially with respect to viruses and other attackers.
Instead, it looks like the real issue might be that clonal reproduction introduces a ratchet where the population can’t clear genetic errors. According to the scientists of the cloning paper:
… mammals rely on sexual rather than asexual reproduction to eliminate genetic anomalies caused by clonal reproduction.
Without sex, the genetic damage just ratchets up and up over a couple dozen generations. It’s sort of like model collapse in LLMs: you train on their outputs, over and over, until the snake eats its tail. Surprisingly, the researchers reported that visually the cloned mice seemed relatively healthy in the earlier generations. But then again, these clones are leading highly sedentary lives: they basically just have to show up, eat, and drink, and that to the researchers is “healthy” because that’s what mice in a cage their whole lives do. The researchers couldn’t tell that under the surface a lot of genetic damage wasn’t shed by sexual reproduction.
But if the ratchet theory is true, we should probably take a closer look at anything that might introduce small compounding errors across generations.
So what about IVF?
IVF now accounts for almost 3% of births in the United States (the highest concentration appears to be almost 10% of births in San Francisco). Of course, IVF is entirely different from cloning, and doesn’t introduce genetic errors. But we do have evidence that the IVF process can create epigenetic damage. While that may sound scary, the first-generation effects of IVF appear relatively small in the grand scheme of things, and the gain of IVF is, of course, basically incalculable (the miracle of an entire human where none would be). So please keep that in mind. But what if the epigenetic damage of IVF compounds generationally? That would be bad.
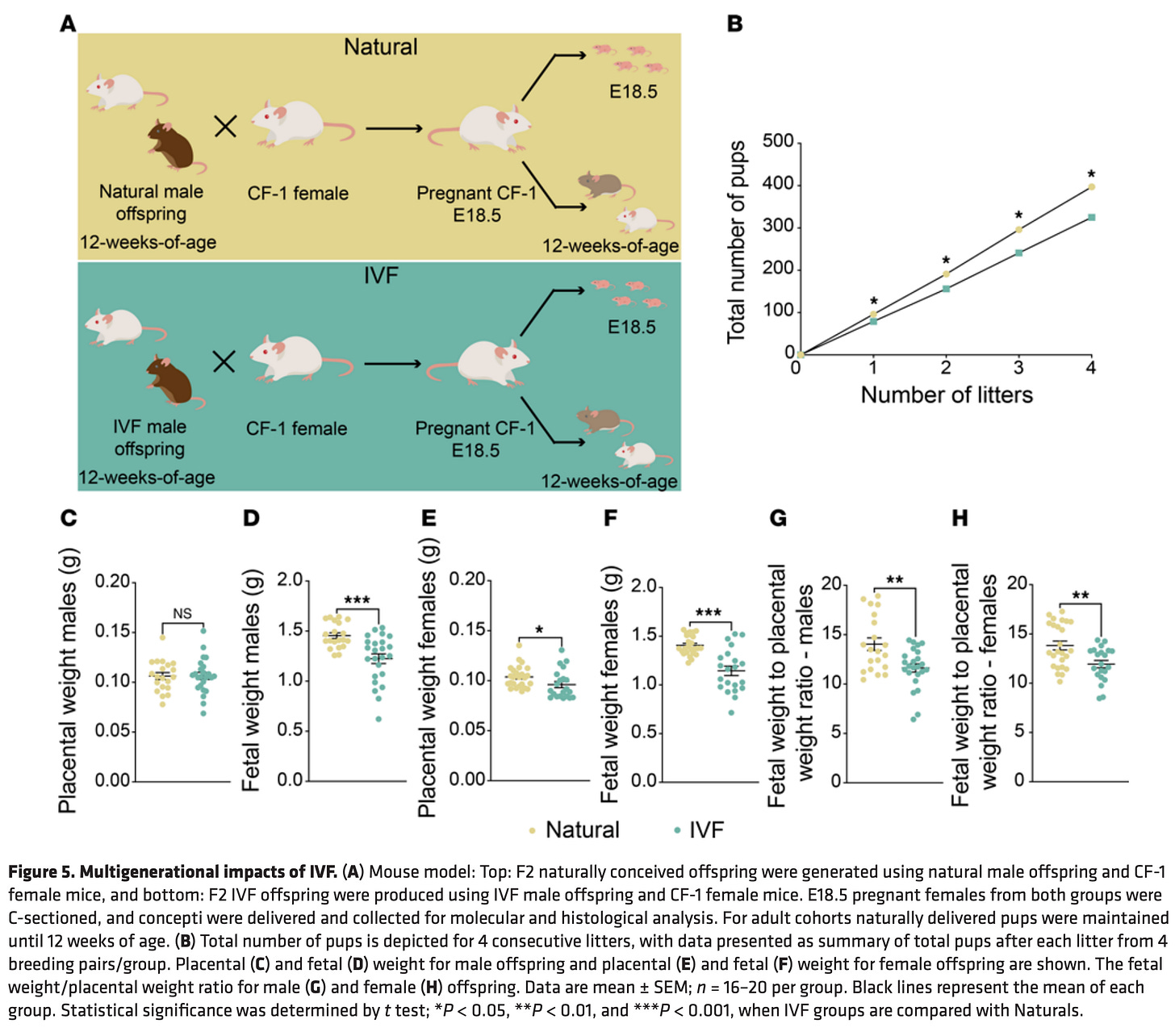
Unfortunately, research indicates this could be true, at least from early mice evidence. In “In vitro fertilization induces reproductive changes in male mouse offspring and has multigenerational effects” the researchers emphasized that:
These findings underscore that the negative effects of IVF not only persist but also may intensify in subsequent generations.
This is not great, because, as the researchers point out:
Although considered safe, IVF pregnancies are associated with an increased risk of perinatal, neonatal, and placental complications; rare genetic syndromes; and possible long-term effects in human and mouse offspring. One possible mechanism for adverse outcomes suggests that IVF procedures occur during critical windows of epigenetic reprogramming in gametes and preimplantation embryos, generating errors that could ultimately affect normal development…. Previous studies have shown that testicular or sperm maturation changes affect normal sperm function, which leads to adverse outcomes in sired offspring.
In other words, IVF can have an effect on the germline, and this could then be passed on via more damage, etc. Which is pretty much what they observe.
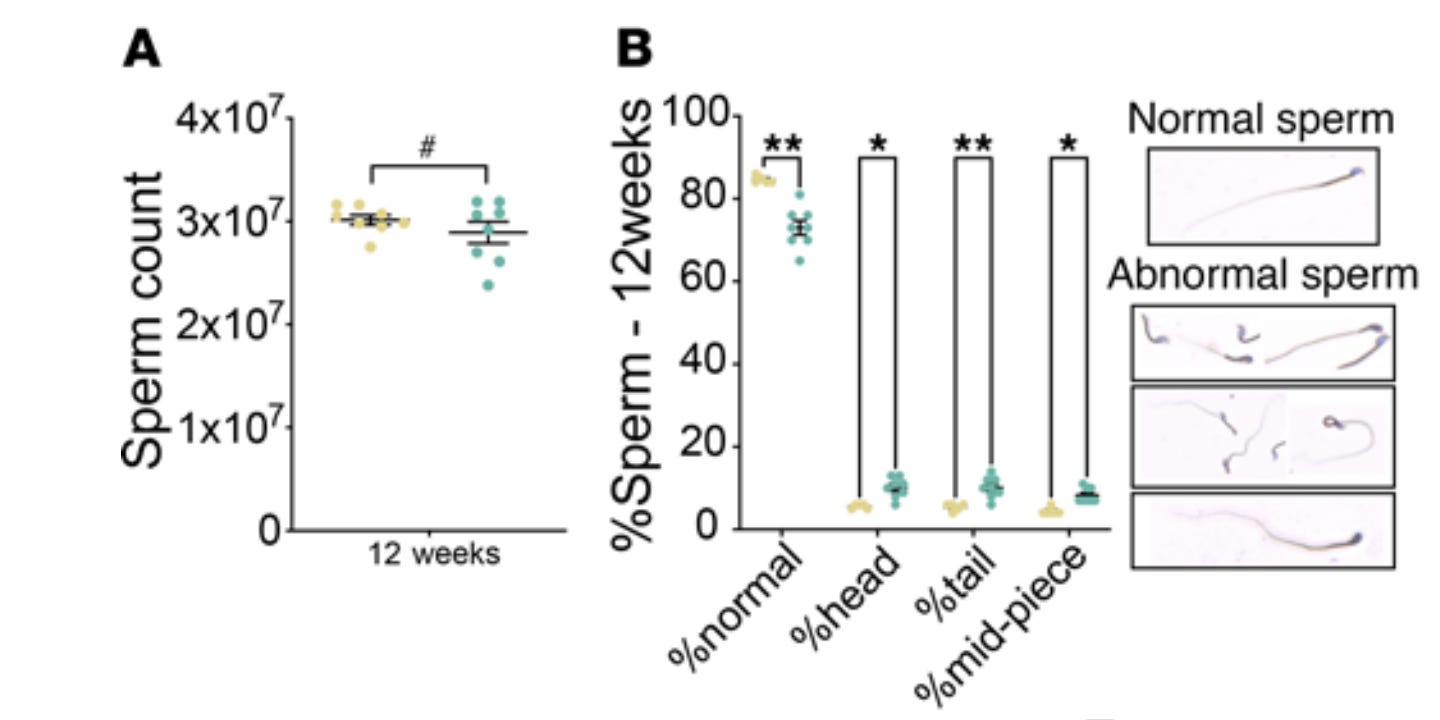
E.g., sperm count in the IVF group is lower (along with testosterone) which then could lead to epigenetic damage being transmitted, since sperm are bearers of epigenetic information, and so on, creating a feedback loop.
What’s also worrying is that the issues spread beyond the germline.
Interestingly, we observed sex-specific adverse outcomes in the F2 from IVF offspring, including a higher risk of insulin and glucose resistance in males and a diabetic phenotype in females. These sex-specific differences can arise by sex-linked genes or hormones. We also observed more severe metabolic and gene expression changes in the F2 generation, as evidenced by the F2 liver RNA-Seq data. These effects could result from the cumulative impact of IVF in the F1 generation, together with a potential contribution of metabolic syndrome in the males, which may be inherited through the germline.
It appears that no one has tracked IVF side effects beyond two generations. That seems kind of important to know!